CML paper summary - February 2026
How I evaluate and treat resistance and relapse in chronic myeloid leukemia
Soverini S & Castagnetti F. Blood, January 2026
Link to full paper: https://ashpublications.org/blood/article-abstract/147/4/337/537310/How-I-evaluate-and-treat-resistance-and-relapse-in?redirectedFrom=fulltext
Summary
This “How I Treat” review outlines a practical framework for evaluating and managing resistance and relapse in chronic myeloid leukemia (CML) in the era of multiple tyrosine kinase inhibitors (TKIs). While most patients achieve excellent long-term outcomes, up to 50% require treatment changes due to resistance or intolerance. The authors emphasise that resistance is a dynamic process that must be interpreted in light of molecular response milestones, mutation status, prior TKI exposure, disease kinetics, comorbidities, and patient goals.
Timely molecular monitoring and appropriate BCR::ABL1 kinase domain mutation testing, preferably by next-generation sequencing (NGS), are central to informed decision-making. Rational, mutation-informed, TKI sequencing is essential to prevent clonal evolution, with ponatinib, asciminib, and allogeneic stem cell transplantation (Allo SCT) reserved for selected later-line settings.
Key points for clinicians
- Resistance should be defined using European LeukemiaNet (ELN) or National Comprehensive Cancer Network (NCCN) molecular milestones, rather than single transcript values.
- Always rule out non-adherence, drug interactions, and dose reductions before declaring true resistance.
- BCR::ABL1 tyrosine kinase domain (TKD) mutation testing is mandatory when resistance or relapse is suspected.
- NGS is preferred over Sanger sequencing for earlier detection and identification of mutations.
- Mutation status should primarily be used to exclude ineffective TKIs rather than to select a single “best” drug.
- Ponatinib and asciminib are preferred after failure of second-generation TKIs, but patient comorbidities and toxicity risks must guide choice and dosing.
- Delayed or inappropriate switching increases the risk of compound mutations and disease progression.
Introduction
The introduction of TKIs has transformed CML from a fatal disease into a chronic condition with near-normal life expectancy for most patients. However, this success can obscure the complexity of managing patients who fail to meet response milestones or who lose a previously achieved response. While only a small proportion progress to advanced phases, resistance or interolerance necessitating a change in therapy occures in up to half of patients over the disease course.
With six approved TKIs spanning different generations and mechanisms of action, including the allosteric inhibitor asciminib, clinicians now have multiple therapeutic options. This abundance requires careful sequencing to maximise efficacy and minimise toxicity. The authors stress that “one-size-fits-all” rules are inappropriate; instead, individualised decision-making based on clinical and biological factors is essential.
This review aims to guide clinicians through:
- How resistance and relapse are defined,
- Which molecular testing should be performed and when,
- How to interpret molecular and cytogenetic findings,
- And how to integrate these data into rational treatment decisions.
Methods
This article is an expert narrative review within the Blood “How I Treat” series and is based on:
- Current ELN and NCCN guidelines for response assessment and resistance definitions.
- Extensive review of clinical trials, real-world studies, and population-based registries.
- Integration of molecular diagnostics, including quantitative PCR, cytogenetics, and mutation testing methodologies.
- Two illustrative clinical cases demonstrating practical application of the decision framework.
Key findings
Defining resistance and relapse
Resistance is defined according to molecular response milestones rather than symptoms or hematologic parameters alone.
- ELN 2020 criteria classify responses as optimal, warning, or failure at 3, 6, and 12 months based on BCR::ABL1 IS levels
- NCCN guidelines use a "traffic-light" system (green/yellow/orange/red) to guide treatment changes.
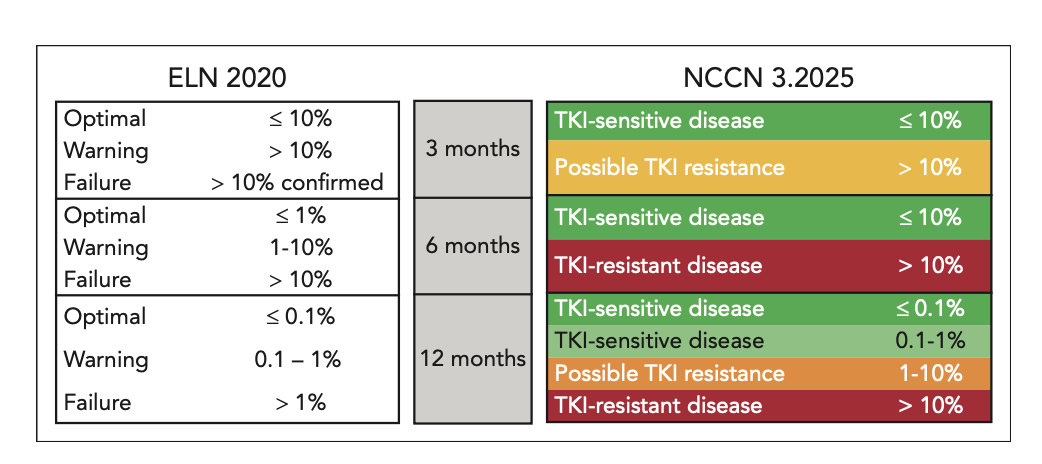
Source: How I evaluate and treat resistance and relapse in CML
Soverini S & Castagnetti F. Blood, January 2026, Figure 1
Loss of response (secondary resistance) is as clinically significant as failure to achieve milestones (primary resistance). Reliable molecular monitoring every 3 months until stable MMR is essential to detect early relapse.
First step: exclude extrinsic causes
Before labeling resistance as biologic, clinicians must exclude:
- Non-adherence, a common and under-recognised issue in chronic therapy.
- Drug-drug interactions, particularly in older patients with polypharmacy.
- Dose reductions or interruptions due to toxicity that may compromise efficacy.
Addressing these factors can sometimes restore response without changing therapy.
Role of BCR::ABL1 TKD mutation testing
Mutation testing is mandatory when resistance or relapse is suspected.
Figure: Proposed algorithm to use BCR::ABL1 TKD mutation results in clinical decision-making
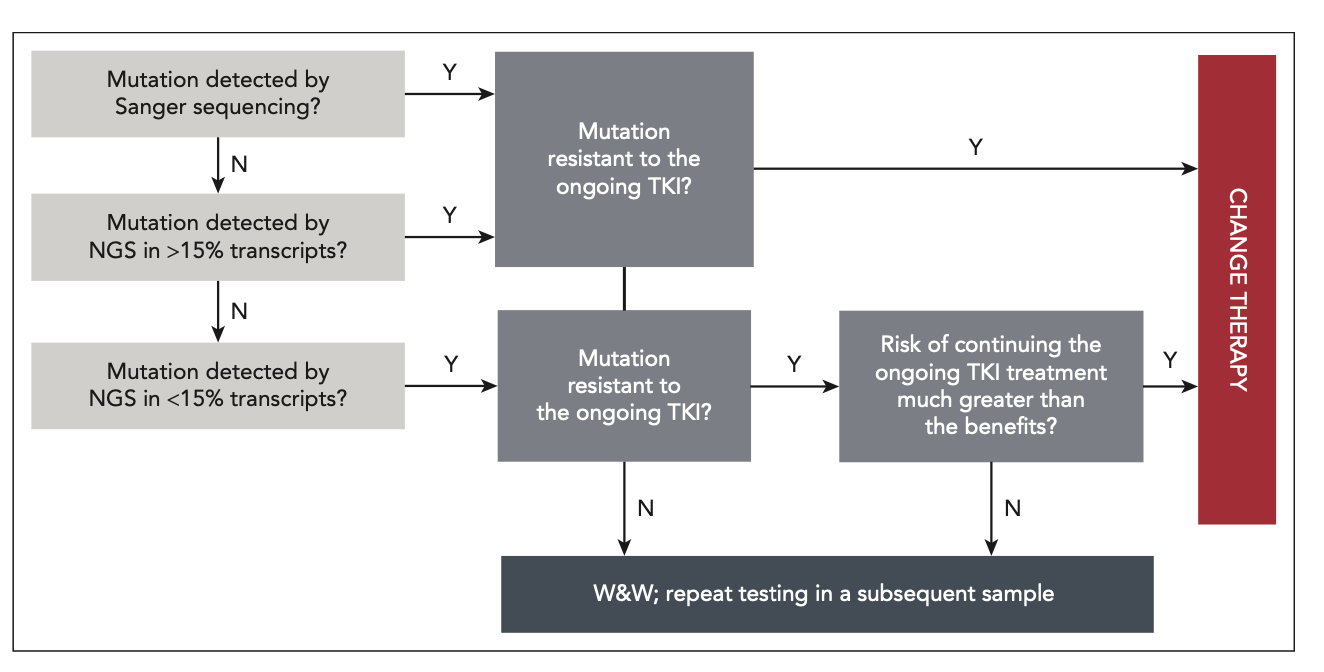
Source: How I evaluate and treat resistance and relapse in CML
Soverini S & Castagnetti F. Blood, January 2026, Figure 2
- NGS is preferred due to higher sensitivity and ability to detect low-level and compound mutations.
- Sanger sequencing remains acceptable but may miss emerging clones.
- Digital PCR can be useful for targeted detection (e.g., T315I).
Key principles:
- Mutation status should guide which TKIs must be avoided.
- Compound mutations (especially T315I-inclusive) are associated with high-level resistance and limited therapeutic options.
- Delayed switching increases the risk of acquiring compound mutations.
Additional cytogenetic and molecular considerations
- Additional chromosomal abnormalities (ACAs), particularly high-risk ACAs, may signal disease progression and warrant therapy change.
- Non-BCR::ABL1 mutations (e.g., ASXL1) are increasingly recognised, but their role in guiding therapy remains investigational. Routine testing is not yet recommended outside research settings.
Choosing the next TKI: a structured approach
Treatment selection should consider:
- TKIs previously used and their doses
- Mutation profile
- BCR::ABL1 transcript level and kinetics
- Patient goals (e.g., treatment-free remission)
- Age and comorbidities, especially cardiovascular risk
General sequencing principles:
- After imatinib failure→ use a second-generation TKI.
- After second-generation TKI failure→ prefer ponatinib or asciminib.
- Ponatinib is preferred for T315I-positive disease but requires careful cardiovascular risk assessment and dose optimisation.
- Asciminib is particularly valuable for patients with intolerance or comorbidities due to its favorable safety profile.
Role of allogeneic stem cell transplantation
Allo-SCT should be considered in:
- Patients resistant or intolerant to ≥2 TKIs,
- Patients with compound mutations resistant to available TKIs,
- Patients with disease progression or inadequate hematologic recovery.
While less frequently used today, transplantation remains a potentially curative option in selected cases.
Case-based practical insights
The two cases illustrate:
- Rational selection of asciminib in a patient without mutations but with comorbidities limiting other TKIs.
- Dose-dependent management of ponatinib in T315I-positive disease, balancing molecular control with cardiovascular risk.
Conclusions
Management of resistance and relapse in CML requires individualised, evidence-based decision-making that integrates molecular data, treatment history, disease kinetics, and patient characteristics. Early recognition of suboptimal response, timely mutation testing, and rational TKI sequencing are essential to prevent disease progression and clonal evolution.
Ponatinib and asciminib have expanded therapeutic possibilities for multi-resistant patients, while allo-SCT remains relevant for selected high-risk cases. As new TKIs and combination strategies emerge, future management will increasingly rely on precise molecular characterisation and personalised treatment approaches.
For clinicians, this review provides a clear roadmap for navigating resistance in CML, emphasising that careful, informed choices can still achieve durable disease control even after multiple lines of therapy.










